Cellular organelles called mitochondria prepare the fuel for most biochemical reactions in our body by a process called respiration, by which oxygen is reacted with electrons derived from food. A team from Department of Biochemistry and Biophysics, Stockholm University led by Professor Martin Ott studies how the respiratory chain complexes are put together. This is a complicated process because these complexes are build up from many protein subunits.
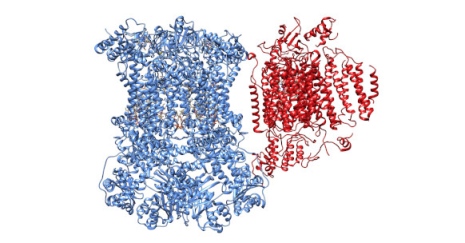
Martin Ott and his research team have now used electron cryo-microscopy to determine the first high-resolution structures of the yeast supercomplex in which the two terminal enzymes of cellular respiration interact. This work was entirely undertaken at the new electron cryo-microscopy facility at the Science for Life Laboratory in Stockholm, lead by Marta Carroni and Julian Conrad. The study is published in the journal Nature Structural and Molecular Biology.
Organization of subsequent chemical reactions in large enzyme assemblies
Cellular respiration is an essential process of life, as it allows a very efficient use of the chemical energy stored in food. The enzymes mediating this process are multi-subunit assemblies with a very complex architecture, which extract chemical energy of nutrients in a step-wise manner through the transfer of electrons. It was previously suggested that the complexes are independent units connected by mobile electron carriers, but biochemical work 20 years ago demonstrated that these complexes instead form large, supramolecular assemblies called respiratory supercomplexes. “However, the precise structure of the individual complexes or the chemical nature of their interactions was not known prior to our study”, says Sorbhi Rathore at Department of Biochemistry and Biophysics, Stockholm University and lead author of the study.
With the aid of their 3D reconstruction of the respiratory supercomplex, she and her colleagues at the department were able to show that the enzymes are positioned such that the binding sites of the mobile electron carrier cytochrome c are positioned to facilitate electron transport. This is made possible by the fact that the two complexes have evolved to contain interaction surfaces that bring the two partners in close and well-defined proximity. Moreover, the interaction sites are build up by a tight protein-protein interaction and through additional phospholipid bridges, which tethers the complexes at the level of the mitochondrial membrane.
"Why respiratory chain complexes assemble into supercomplexes has so far been unknown. Because of the central role of mitochondrial energy conversion for the functioning of cells and organisms, this has been a central question in the field. Our structure now provides us with the information necessary to disrupt the interaction through mutations and then studying the consequences,” says Jens Berndtsson, another lead author of the study.
Stockholm University early with own electron cryo-microscopy
Six years ago, electron cryo-microscopy made a big leap forward to allow obtaining high-resolution structures of protein complexes, a breakthrough awarded with the Nobel prize in Chemistry in 2017. Stockholm University was among the first universities in Europe to establish its own facility that is up and running since two years.
“These novel structures are really exciting as they allow us to inspect for the first time these highly complex molecular machineries. These structures really tell a lot about how these machines work and how they are build, a process that is so important for life," says Professor Martin Ott at Department of Biochemistry and Biophysics, Stockholm University.
More information: Sorbhi Rathore, Jens Berndtsson, Lorena Marin Buera et al. Cryo-EM structure of the yeast respiratory supercomplex, Nature Structural & Molecular Biology (2018). DOI: 10.1038/s41594-018-0169-7
Read more about Martin Ott, Professor of Biochemistry/Wallenberg Academy Fellow at Department of Biochemistry and Biophysics, Stockholm University

