Capturing protein dynamics in crowded environments with European XFEL
Proteins are responsible for many of the body’s functions and understanding protein dynamics in cells enables scientists to learn more about drug design and disease pathology. Now, an international research team has identified a new way of probing protein dynamics using the high repetition rate and coherence of the X-ray flashes produced by the European X-ray Free-Electron Laser (EuXFEL). The study, published in Nature Communications, uses a technique called megahertz X-ray photon correlation spectroscopy (MHz-XPCS) and could open new applications in health and pharmaceuticals.
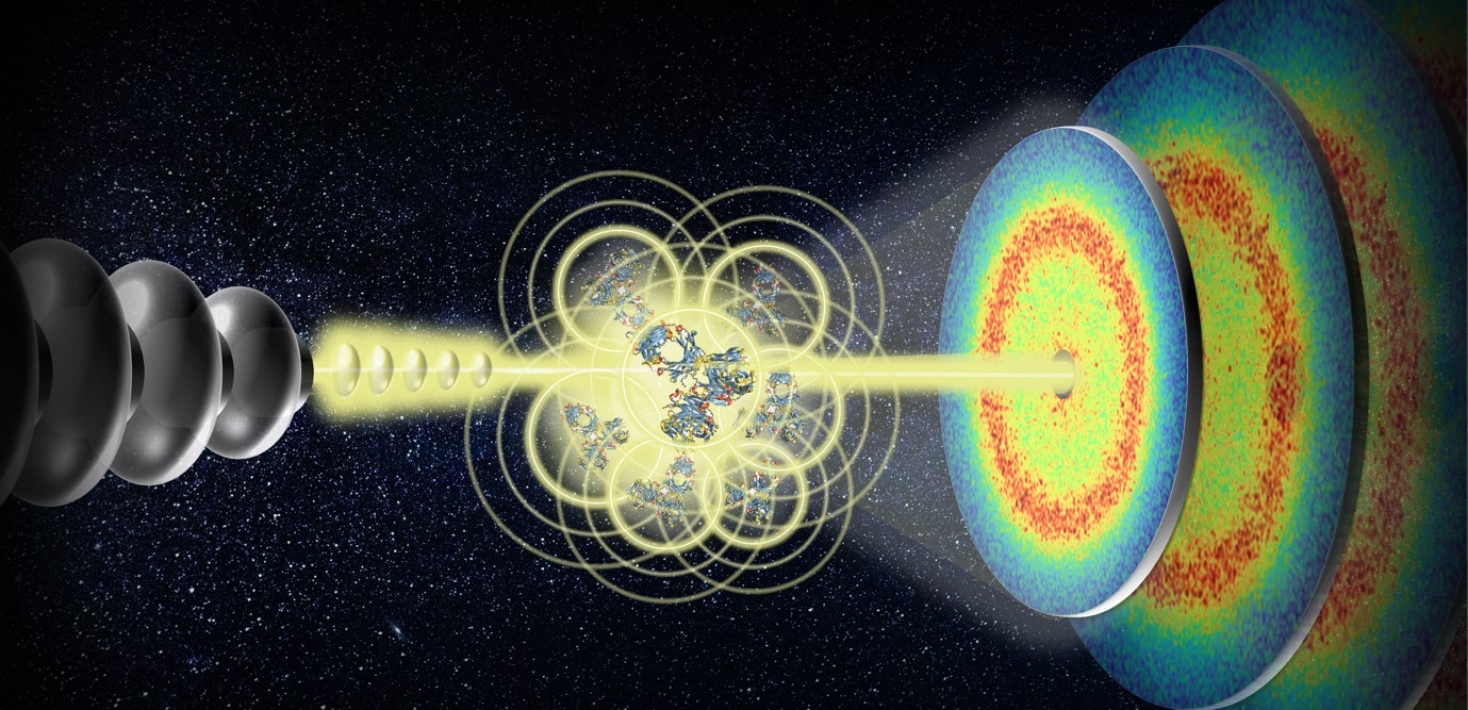
The study was led by Stockholm University, the University of Tübingen, the University of Siegen as well as EuXFEL and used the Materials Imaging and Dynamics (MID) instrument at EuXFEL to examine antibody protein solutions. The X-ray laser produces millions of flashes per second, enabling scientists to investigate proteins on time scales shorter than a microsecond and on length scales as small as a molecule.
Damaging proteins or causing them to react
Typically, the combination of high pulse rates and high energy radiation can damage proteins or cause them to react. But by lowering the X-ray radiation dose and frequently moving the protein samples, the researchers have identified a way of probing the dynamics of the proteins before damage sets in, alongside measuring the impact of the X-ray beam on the motion of the proteins.
“Low-dosage MHz-XPCS is a method that allows us to examine protein dynamics in ways that were previously impossible,” says co-author on the study Anders Madsen, leading scientist at EuXFEL and in charge of the MID instrument. “By combining the high pulse rate of the European XFEL with a reduced radiation dosage, we can examine new behaviors in these proteins”.
“The reactions of proteins on small scales, microsecond by microsecond, are closely related to interactions that determine a number of their properties,” Madsen adds. “This can help scientists to design new, more modern drugs as well as improving our understanding of protein-related diseases.”
Using MHz-XPCS, researchers can now also investigate the collective behaviour of many protein molecules at once. To do this, the team used highly concentrated bovine immunoglobulin protein solution that mimics the crowded and complex environment of a cell.
“Many factors influence proteins within the crowded environment of our cells, all of which slow down and hinder protein movement,” says Prof. Foivos Perakis from Stockholm University. “Studies also show that proteins in crowded cellular spaces behave differently to those of other systems. All these factors need to be considered for a full understanding of these systems.”
Fresh insights into antibody proteins
The study has not only opened a new pathway of investigations into protein dynamics - it has also delivered fresh insights into antibody proteins themselves, including showing how the diffusion of proteins in a solution changed with the applied radiation dose.
“When drugs enter our bloodstream, they bind to proteins that transport and distribute them around the body,” explains Prof. Christian Gutt, corresponding author from the University of Siegen in Germany. “A comprehensive understanding of these kinds of processes could lead to more effective treatments with fewer side effects.”
The team now intends to use their new, low-dosage MHz-XPCS technique to fill in the gap in our knowledge of proteins, where previous measurement techniques could not be used.
“X-ray photon correlation spectroscopy, combined with the high repetition rate of the European XFEL, can access these relevant measurement parameters,” concludes Dr Fajun Zhang, scientist at the University of Tübingen in Germany. “Importantly, it can provide information on processes that we have limited understanding of, such as cluster formation.”
Reference
Resolving molecular diffusion and aggregation of antibody proteins with megahertz X-ray free-electron laser pulses
M. Reiser*, A. Girelli, A. Ragulskaya, S. Das, S. Berkowicz, M. Bin, M. Ladd-Parada, M. Filianina, H.-F. Poggemann, N. Begam, M. S. Akhundzadeh, S. Timmermann, L. Randolph, Y. Chushkin, T. Seydel, U. Boesenberg, J. Hallmann, J. Möller, A. Rodriguez-Fernandez, R. Rosca, R. Schaffer, M. Scholz, R. Shayduk, A. Zozulya, A. Madsen, F. Schreiber, F. Zhang, F. Perakis* and C. Gutt* [*corresponding authors] Nature Comm. (2022) DOI:
More information
- Foivos Perakis
- Research Group SDAQS
-
Nat. Commun. 13, 5528 (2022) https://doi.org/10.1038/s41467-022-33154-7
Last updated: October 10, 2022
Source: Fysikum