Study opens for extended use of lignin in sustainable chemistry
Researchers at Stockholm University have found a new cheap and environmentally-friendly oxidant that is highly efficient in cleaving bonds in lignin at low temperatures. Extended use of lignin has the potential to replace many chemicals that we today get from fossil sources.
Lignin is the difference between wood and paper and the part that is liberated during pulping. Today, this part of the wood is mainly burnt to a low value during the process of producing pulp for paper, boxes, and textile fibers. This is of course not sustainable and lignin has the potential to replace all aromatic chemicals that we today get from fossil sources. Thus, valorizing lignin is an important goal in the transformation to a bio-based society. A new research direction has emerged where novel pulping methods involve both generating the high-value pulp as well as producing renewable chemicals from the lignin.
Catalytic methods that can cleave more bonds
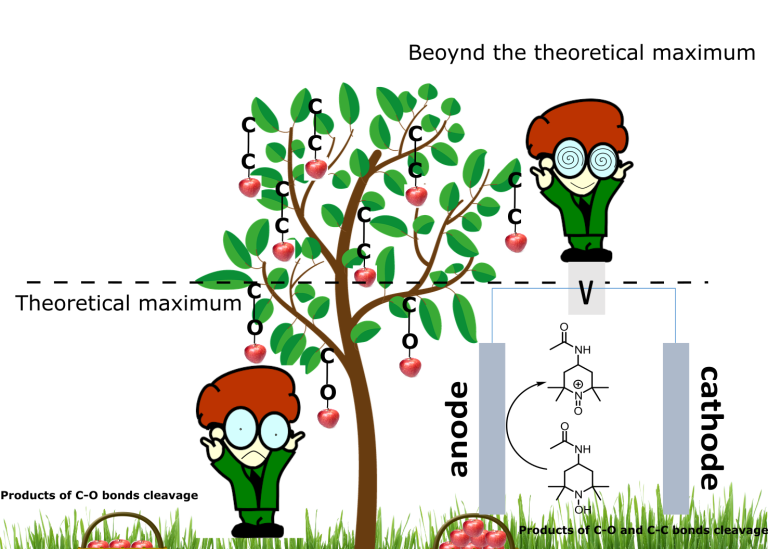
Lignin is a complex aromatic polymer with various chemical bonds. Over the past decade, there has been massive development in cleaving carbon–oxygen bonds which are relatively easy to cleave by a catalyst. These easy bonds ‘the low hanging fruit’ comprise about 60-70 percent of the chemical bonds in lignin. If all these bonds are cleaved ‘the theoretical maximum yield’ of monomers is between 36 percent and 49 percent. A research group at Stockholm University has previously developed catalytic methods that can cleave above 90 percent of all these bonds.
Cheap and environmentally-friendly oxidant
Thus, the existing methods give less than 50 percent yield of high-value compounds from lignin. To utilize the raw material better, new approaches targeting other chemical bonds in lignin are required. However, as these bonds are hard to cleave, this has so far only been successful using very harsh reaction conditions. Furthermore, these reactions give a wide range of products with poor selectivity.

“We found a very cheap and environmentally-friendly oxidant that is highly efficient in cleaving several of the recalcitrant carbon–carbon bonds in lignin at low temperatures. In a Nature Chemistry article we show that the oxidant can be easily regenerated by electrochemistry, which is benign if green electricity is used. What is noteworthy is that we selectively generate 2,6-dimethoxybenzoquinone which is an important substance in the chemical industries,” says Joseph Samec, Professor of Organic Chemistry and corresponding author of the article.
Sustainable chemistry applied to solve difficult challenges
The results have proven that green and sustainable chemistry can be applied to solve difficult challenges, here on lignin depolymerization, and moves us closer to more efficient value chains using biomass as raw material. “Our goal is to develop novel sustainable routes to generate various essential chemicals from low-value residues and wastes based on green chemistry,” says Joseph Samec.
Read article Oxidative cleavage of C–C bonds in lignin in Nature Chemistry
Read more on Joseph Samec´s research
Last updated: September 23, 2021
Source: Communications Office

