Michael OdeliusProfessor
About me
With a background in physical chemistry, I work with research, teaching and out reach projects at the Department of Physics. I study chemical processes and molecules using computer simulations based on quantum chemical calculations.
Teaching
At the department of physics at Stockholm University, I have lectured in courses on wave dynamics for teachers at the Bachelor level and simulation methods in statistical physics within the master programme in computational physics. I am presently responsible for the course on quantum chemistry, which we recently converted into flipped class room format. Previous teaching experience is in the field of chemistry, in particular physical chemistry and computational chemistry.
If you are interested in a bachelor och Master project, please feel welcome to contact me. There are several examples of previous projects on the home page of the division of chemical physics.
In addition, I am strongly engaged in out reach activities for schools, high-schools and the general public. In particular, Fysikum has a strong involvement in European Researcher's Night in Stockholm, coordinated by the House of Science, at AlbaNova university centre.
Research
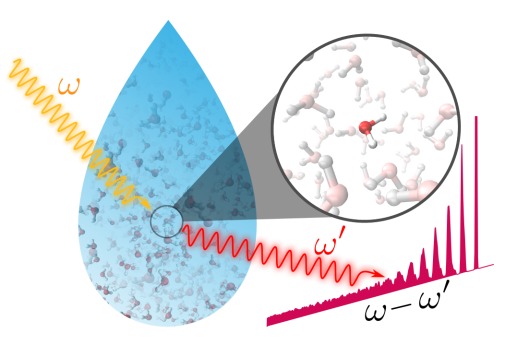

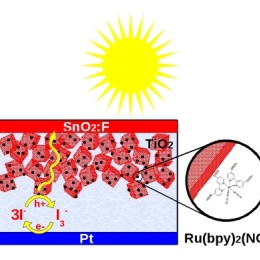
We use theoretical calculations based on quantum mechanics and statistical physics to study solutions and and solar cells. These methods allow us to describe the electronic structure and chemical dynamics on a molecular level. Through computer simulations, we can, in close collaboration with experimental groups, reach a detailed insight into complicated systems and for example contribute to the development of more efficient chemical storage of solar energy.
On the home page of the research group, you can learn more about on-going research projects and find examples of recent publications.
Job Opportunity
We are currently looking for a candidate for a PhD studentship in Theoretical Chemical Physics for investigations of energy-rich azide compounds. The photochemistry of energy-rich azides (R-N=N=N) shows a great diversity in organic synthesis and in the important field of click chemistry, which was awarded the Nobel prize in Chemistry 2022.
Research projects
Publications
A selection from Stockholm University publication database
-
Electronic Structure Changes of an Aromatic Amine Photoacid along the Förster Cycle
2022. Sebastian Eckert (et al.). Angewandte Chemie International Edition 61 (27)
ArticleRead more about Electronic Structure Changes of an Aromatic Amine Photoacid along the Förster CyclePhotoacids show a strong increase in acidity in the first electronic excited state, enabling real-time studies of proton transfer in acid-base reactions, proton transport in energy storage devices and biomolecular sensor protein systems. Several explanations have been proposed for what determines photoacidity, ranging from variations in solvation free energy to changes in electronic structure occurring along the four stages of the Forster cycle. Here we use picosecond nitrogen K-edge spectroscopy to monitor the electronic structure changes of the proton donating group in a protonated aromatic amine photoacid in solution upon photoexcitation and subsequent proton transfer dynamics. Probing core-to-valence transitions locally at the amine functional group and with orbital specificity, we clearly reveal pronounced electronic structure, dipole moment and energetic changes on the conjugate photobase side. This result paves the way for a detailed electronic structural characterization of the photoacidity phenomenon.
-
A-site cation influence on the conduction band of lead bromide perovskites
2022. Gabriel J. Man (et al.). Nature Communications 13 (1)
ArticleRead more about A-site cation influence on the conduction band of lead bromide perovskitesHot carrier solar cells hold promise for exceeding the Shockley-Queisser limit. Slow hot carrier cooling is one of the most intriguing properties of lead halide perovskites and distinguishes this class of materials from competing materials used in solar cells. Here we use the element selectivity of high-resolution X-ray spectroscopy and density functional theory to uncover a previously hidden feature in the conduction band states, the sigma-pi energy splitting, and find that it is strongly influenced by the strength of electronic coupling between the A-cation and bromide-lead sublattice. Our finding provides an alternative mechanism to the commonly discussed polaronic screening and hot phonon bottleneck carrier cooling mechanisms. Our work emphasizes the optoelectronic role of the A-cation, provides a comprehensive view of A-cation effects in the crystal and electronic structures, and outlines a broadly applicable spectroscopic approach for assessing the impact of chemical alterations of the A-cation on perovskite electronic structure. The A-cation influence on the mechanism of slow hot carrier cooling in perovskites is controversial. Here, Man et al. resolve a debated issue regarding A-cation influence on the electronic structure of lead halide perovskites.
-
Spatial microheterogeneity in the valence band of mixed halide hybrid perovskite materials
2022. Axel Erbing (et al.). Chemical Science 13 (32), 9285-9294
ArticleRead more about Spatial microheterogeneity in the valence band of mixed halide hybrid perovskite materialsThe valence band of lead halide hybrid perovskites with a mixed I/Br composition is investigated using electronic structure calculations and complementarily probed with hard X-ray photoelectron spectroscopy. In the latter, we used high photon energies giving element sensitivity to the heavy lead and halide ions and we observe distinct trends in the valence band as a function of the I : Br ratio. Through electronic structure calculations, we show that the spectral trends with overall composition can be understood in terms of variations in the local environment of neighboring halide ions. From the computational model supported by the experimental evidence, a picture of the microheterogeneity in the valence band maximum emerges. The microheterogeneity in the valence band suggests that additional charge transport mechanisms might be active in lead mixed halide hybrid perovskites, which could be described in terms of percolation pathways.
-
Photoinduced bond oscillations in ironpentacarbonyl give delayed synchronous bursts of carbonmonoxide release
2022. Ambar Banerjee (et al.). Nature Communications 13 (1)
ArticleRead more about Photoinduced bond oscillations in ironpentacarbonyl give delayed synchronous bursts of carbonmonoxide releaseEarly excited state dynamics in the photodissociation of transition metal carbonyls determines the chemical nature of short-lived catalytically active reaction intermediates. However, time-resolved experiments have not yet revealed mechanistic details in the sub-picosecond regime. Hence, in this study the photoexcitation of ironpentacarbonyl Fe(CO)5 is simulated with semi-classical excited state molecular dynamics. We find that the bright metal-to-ligand charge-transfer (MLCT) transition induces synchronous Fe-C oscillations in the trigonal bipyramidal complex leading to periodically reoccurring release of predominantly axial CO. Metaphorically the photoactivated Fe(CO)5 acts as a CO geyser, as a result of dynamics in the potential energy landscape of the axial Fe-C distances and non-adiabatic transitions between manifolds of bound MLCT and dissociative metal-centered (MC) excited states. The predominant release of axial CO ligands and delayed release of equatorial CO ligands are explained in a unified mechanism based on the σ*(Fe-C) anti-bonding character of the receiving orbital in the dissociative MC states.
-
Core-Level Binding Energy Reveals Hydrogen Bonding Configurations of Water Adsorbed on TiO2 (110) Surface
2021. Chinnathambi Kamal (et al.). Physical Review Letters 126 (1)
ArticleRead more about Core-Level Binding Energy Reveals Hydrogen Bonding Configurations of Water Adsorbed on TiO2 (110) SurfaceUsing x-ray photoelectron spectroscopy of the oxygen 1s core level, the ratio between intact (D2O) and dissociated (OD) water in the hydrated stoichiometric TiO2 (110) surface is determined at varying coverage and temperature. In the submonolayer regime, both the D2O:OD ratio and the core-level binding energy of D2O (Delta BE) decrease with temperature. The observed variations in Delta BE are shown with density functional theory to be governed crucially and solely by the local hydrogen bonding environment, revealing a generally applicable classification and details about adsorption motifs.
Show all publications by Michael Odelius at Stockholm University
$presentationText