His software has taken the life sciences to a new level
The GROMACS computer software developed by Erik Lindahl’s research group to simulate the molecules of life, is used all around the world to study different processes in our cells. He is using the software to solve a medical riddle: how can substances like alcohol and narcotics cause our nerve cells to disfunction?

Erik Lindahl, professor of Biophysics at Stockholm University, has always had a passion for both computers and life sciences. Therefore, as a young doctoral student in theoretical physics, when he was developing models for computer simulation, he chose to look at the fatty membrane around our cells.
“We were calculating how the fatty acids moved, but at that time biochemists regarded that form of science as something the cat dragged in, and with good reason. It was something that only theoreticians were doing,” says Lindahl.
His software is a global success
However, as computers became more powerful, computer modelling also became more faithful to reality. The field boomed, and Erik Lindahl’s software, GROMACS, which simulates the molecules of life, is now often a requirement when a research institution invests in a new supercomputer. Every day, biochemists and molecular biologists use the software, which is free, to visualise and understand processes in living organisms.
“On average, every hour of every day of the year, there is an article published that cites what we have done. So GROMACS laid the foundation for my career,” says Erik Lindahl.
His research group are still developing and improving the software, but are now also using it to solve a problem that has long puzzled doctors: how can small molecules like alcohol, Valium and various narcotics, have such a powerful effect on human nerve cells?
A chance meeting that sent research in a new direction
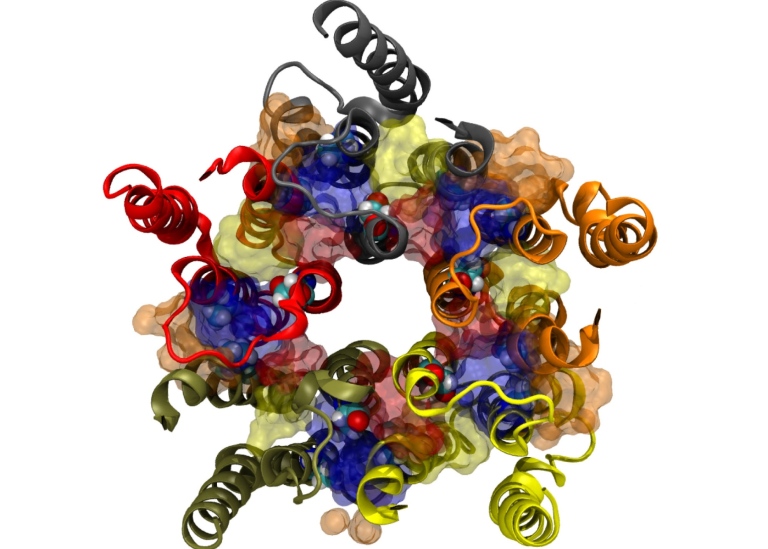
It was chance that led to Erik Lindahl’s interest in the brain. He was a postdoc at Stanford University in California when an anaesthetist, Edward Bertaccini, asked for help. He wondered whether Erik Lindahl could create a computer model of special proteins on the surface of nerve cells, called ion channels. Edward Bertaccini knew that the anaesthetics he gave his patients affected their ion channels, making the nerve cells relax so the patients slept deeply. But how?
“I almost didn’t have time to look at it, but I thought it sounded like an interesting problem,” says Lindahl.
His interest was aroused because ion channels are vital for brain function, as it is via them that nerve cells are activated. Naturally, this is done with the help of signal substances. When a nerve cell sends a signal to another nerve cell, it releases a signal substance that travels across the gap between these cells. This signal substance then latches onto an ion channel on the surface on the nerve cell. It makes the ion channel open up, creating a tiny opening into the nerve cell, through which charged ions can flow and activate the nerve.
Many pharmaceuticals work through ion channels
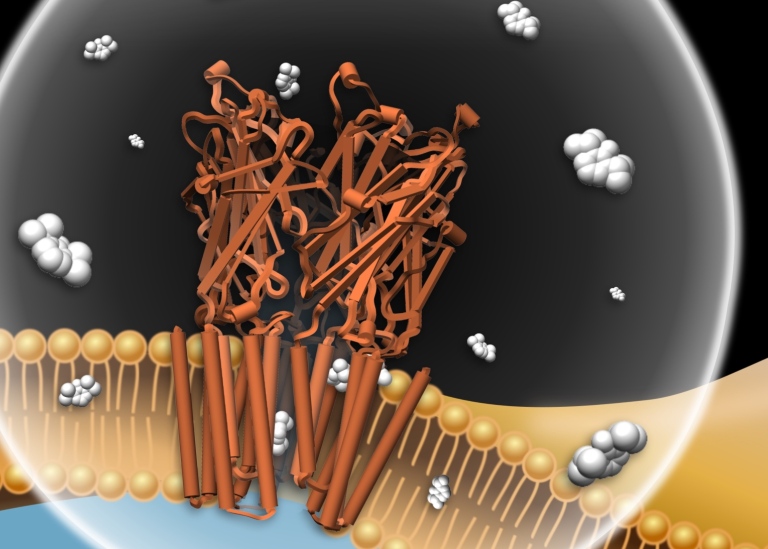
There are lots of different types of ion channels that regulate brain functions, but what they look like and how they work has been terra incognita on the knowledge map.
“We wanted to find out they work, in part so we can develop better neuropharmacological drugs. The ones we currently use are blunt and we don’t know exactly which ion channels they affect,” says Lindahl.
When he started the project, he quickly realised that the research group needed to be able to test whether the computer models they produced were correct. And for this, they needed proper experiments. He therefore added people with lab skills to his research group, and an enthusiastic doctoral student started creating pictures of the ion channels using cryo-electron microscopy, a method that has been rewarded with the Nobel Prize.
The various methods – computer modelling, experiments and microscopy – have created a synergy and, slowly but surely, the research group has gained an understanding of what happens when a narcotic attaches to an ion channel. One surprise has been that the drug does not attach to the part of the ion channel that is on the outside of the nerve cell. Instead, it pushes into the cell’s fatty membrane and interacts with the ion channel there.
“This causes a small change in the in channel that has a huge effect, with enormous movements in the ion channel,” says Lindahl.
The research group has also discovered that different molecules can attach to different places on the ion channels. Some have an activating effect, while others are inhibiting.
“Suddenly, we know how it works, which brings entirely new opportunities for optimising the molecules and adjusting their effects on the ion channels,” he says.
Computer models accelerate progress in the life sciences
Since Erik Lindahl started developing GROMACS, the computers he uses have become about a million times better, revolutionising the life sciences. He takes the coronavirus as an example. Just two years after the start of the pandemic, there are several candidate pharmaceuticals for COVID-19.
“I don’t really think these drugs are fantastic, but we have systematically and rapidly been able to produce drugs for a new disease. It’s happened ten times faster than it usually does,” he says.
Without computer modelling, this would not have been possible.
However, he points out that the computer models still have many weaknesses and there is a lot left to develop.
“But if we can learn to master these types of tools, they are incredibly powerful. They’ll just get better and better,” says Erik Lindahl.
Read more on Erik Lindahl´s research.
Text: Ann Fernholm
Last updated: March 29, 2022
Source: Communications Office

