Eva HedlundProfessor of Neurochemistry
Research
Neurodegenerative diseases are characterized by the selective loss of specific neuronal populations with corresponding distinct clinical features. Why specific neuron types are selectively vulnerable to a neurodegenerative disease process, such as motor neurons in amyotrophic lateral sclerosis (ALS) or dopamine neurons in Parkinson’s disease (PD), is currently unclear. It is particularly intriguing as disease-causative genes are often broadly expressed in the nervous system and sometimes even in every cell in our body.
We are focused on understanding mechanisms of selective neuronal vulnerability and resilience seen in neurodegenerative diseases, with particular emphasis on the lethal motor neuron disease ALS. We believe that investigating cell intrinsic properties of neurons that show either extreme vulnerability or particular resilience and even regenerative properties in response to disease could reveal mechanisms that can be targeted by gene therapy approaches to render neurons more resistant.
Towards this purpose we have developed a method (LCM-seq) that allows for spatial RNA sequencing at the single cell level of partially degraded human tissues with exceptional sensitivity and gene detection. We are now using LCM-seq to unravel motor neuron diversity in the human spinal cord as well as to deduce the response of individual neuron to disease as these are either degenerating, persisting or regenerating in response to ALS.
In several neurodegenerative diseases, it is the long neuronal processes (axons) and their synapses (with other neurons or muscle) that first show signs of pathology and degenerate. Still, these axonal processes are often overlooked. Towards the goal of understanding early disease processes in axons and identify targets for disease intervention, we have developed a highly robust method (Axon-seq) to sequence the content of axons and to analyze disease-induced dysregulation of the axonal mRNA content
We are now utilizing Axon-seq to increase the understanding of axon biology in general and to unravel early disease mechanisms in ALS using neurons specified from human induced pluripotent stem cells where we have introduced disease-causing mutations using CRISPR-Cas9 genome editing.
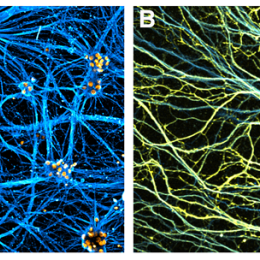
(A) Human motor neurons derived from induced pluripotent stem cells. (B) Human stemcell-derived motor axons grown in isolation from the cell bodies using a microfludic chamber.
Recent publications from the lab:
Correia JC, Kelahmetoglu Y, Jannig PR, Schweingruber C, Svaikovskaya D, Zhengye L, Cervenka I, Khan N, Stec M, Oliveira M, Nijssen J, Martínez-Redondo V, Ducommun S, Azzolini M, Lanner JT, Kleiner S, Hedlund E and Ruas JL (2021) Muscle-secreted neurturin couples fiber oxidative metabolism and slow motor neuron identity. Cell Metabolism 33:2215-2230.
Aguila J, Cheng S, Kee N, Cao M, Wang M, Deng Q* and Hedlund E* (2021) Spatial RNA sequencing identifies robust markers of vulnerable and resistant human midbrain dopamine neurons and their expression in Parkinson's Disease. * corresponding authors. Frontiers in Molecular Neuroscience 14:699562
Nichterwitz S, Nijssen J, Storvall H, Schweingruber C, Comley LH, Allodi I, van der Lee M, Deng Q, Sandberg R and Hedlund E (2020) LCM-seq reveals unique transcriptional adaptation mechanisms and protective pathways of resistant neurons in spinal muscular atrophy. Genome Research 30:1083-1096.
Nizzardo M, Taiana M, Rizzo F, Aguila Benitez J, Nijssen J, Allodi I, Melzi V, Bresolin N, Comi GP, Hedlund E*,# and Corti S*,# (2020) Synaptotagmin 13 is neuroprotective across motor neuron diseases. * corresponding author, # equal contribution. Acta Neuropathologica 139(5):837-853.
Allodi I, Nijssen J, Aguila Benitez J, Schweingruber C, Fuchs A, Bonvicini G, Cao M, Kiehn O and Hedlund E (2019) Modeling motor neuron resilience in ALS using stem cells. Stem Cell Reports 12(6):1329-1341.
Nijssen J, Aguila Benitez J, Hoogstraten R, Kee N and Hedlund E (2018) Axon-seq decodes the motor axon transcriptome and its modulation in response to ALS.Stem Cell Reports 11(6):1565-1578.
Nichterwitz S1, Chen G1, Aguila Benitez J, Storvall H, Yilmaz M, Cao M, Sandberg R, Deng Q2* and Hedlund E2* (2016) Laser-capture microscopy coupled with Smart-seq2 (LCM-seq) for precise spatial transcriptome profiling. 1,2 equal contribution, * corresponding authors. Nature Communications 7:12139
Allodi I1, Comley L1, Nichterwitz S1, Nizzardo M, Simone C, Aguila Benitez J, Cao M, Corti S2* and Hedlund E2* (2016) Differential neuronal vulnerability identifies IGF-2 as a protective factor in ALS. 1,2 equal contribution, * corresponding authors Scientific Reports 6:25960
Group members
Susanne Nichterwitz, Researcher
Christoph Schweingruber, Researcher
Salim Benlefki, Researcher
Nigel Kee, Postdoc
Melanie Leboeuf, Postdoc
Silvia Gomez Alcalde, PhD student
Irene Mei, PhD student
Research projects
$presentationText